Aspectos básicos del (MCP) Modelo Cinético de Partículas:
Aprendizaje esperado:
Describe los aspectos básicos que conforman el modelo cinético de partículas y explica el efecto de la velocidad de éstas.
Las partículas son
*microscópicas
*indivisibles,
*con masa,
*movimiento,
*interacciones y
*vacío entre ellas.
History of Atomic Structure
The search for the atom began as a philosophical question. It was the natural philosophers of ancient Greece that began the search for the atom by asking such questions as: What is stuff composed of? What is the structure of material objects? Is there a basic unit from which all objects are made? As early as 400 B.C., some Greek philosophers proposed that matter is made of indivisible building blocks known as atomos. (Atomos in Greek means indivisible.) To these early Greeks, matter could not be continuously broken down and divided indefinitely. Rather, there was a basic unit or building block that was indivisible and foundational to its structure. This indivisible building block of which all matter was composed became known as the atom.
The early Greeks were simply philosophers. They did not perform experiments to test their theories. In fact, science as an experimental discipline did not emerge as a credible and popular practice until sometime during the 1600s. So the search for the atom remained a philosophical inquiry for a couple of millennia. From the 1600s to the present century, the search for the atom became an experimental pursuit. Several scientists are notable; among them are Robert Boyle, John Dalton, J.J. Thomson, Ernest Rutherford, and Neils Bohr.
KINETIC PARTICLES MODEL
The kinetic theory of matter (particle theory) says that all matter consists of many, very small particles which are constantly moving or in a continual state of motion.
The degree to which the particles move is determined by the amount of energy they have and their relationship to other particles.
The particles might be atoms, molecules or ions. Use of the general term 'particle' means the precise nature of the particles does not have to be specified.
Particle theory helps to explain properties and behaviour of materials by providing a model which enables us to visualise what is happening on a very small scale inside those materials. As a model it is useful because it appears to explain many phenomena but as with all models it does have limitations.
Diffusion
Diffusion gives us more evidence for our kinetic model
Diffusion is the spreading out of particles when they are free to move in a fluid (liquid or gas).
Task:
Observe the experiments in Physics for you pp17, and explin how they give us evidence for molecular movement in liquids and gases.
The model can be used to help explain:
1. The properties of matter
3. Heat & Temperature phenomena
Pressure phenomena
What happens during physical changes such as melting, boiling and evaporating
3 States of Matter
SOLID
In solids the particles are close together and have limited motion.
In solids the particles:
- are held tightly and packed fairly close together - they are strongly attracted to each other
- are in fixed positions but they do vibrate
LIQUID
In a liquid some of the attraction between particles is overcome which allows the particles more freedom of movement.
In liquids the particles:
- are fairly close together with some attraction between them
- are able to move around in all directions but movement is limited by attractions between particles
GAS
In a gas particles attraction between particles is minimized and the particles move freely throughout the container.
In gases the particles:
-have little attraction between them
- are free to move in all directions and collide with each other and with the walls of a container and are widely spaced out
Postulates of KPM
(i) Every gas consists of a large number of small particles called molecules moving with very high velocities in all possible directions.
(ii) The volume of the individual molecule is negligible as compared to the total volume of the gas.
(iii) Gaseous molecules are perfectly elastic so that there is no net loss of kinetic energy due to their collisions.
(iv) The effect of gravity on the motion of the molecules is negligible.
(v) Gaseous molecules are considered as point masses because they do not posses potential energy. So the attractive and repulsive forces between the gas molecules are negligible.
(vi) The pressure of a gas is due to the continuous bombardment on the walls of the containing vessel.
(vii) At constant temperature the average K.E. of all gases is same.
(viii) The average K.E. of the gas molecules is directly proportional to the absolute temperature.
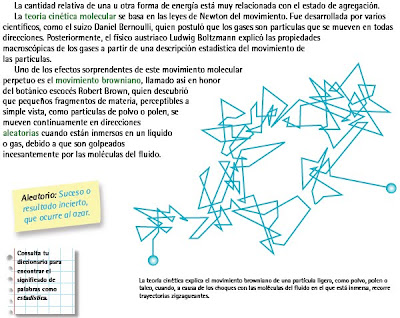
Clausius, Maxwell y Boltzmann
Kinetic theory of gases was developed by Clausius, Maxwell and Boltzmann etc. and represents dynamic particle or microscopic model for different gases since it throws light on the behaviour of the particles (atoms and molecules) which constitute the gases and cannot be seen.
REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW REVIEW
COLEGIO ANDES DE MAZATLÁN SECCIÓN SECUNDARIA
CIENCIAS 2 (FÍSICA) SEGUNDO GRADO
QUIZ 18. EL MODELO CINÉTICO DE PARTÍCULAS
Alumn@: ___________________________________________ Calif.: _____
1. De los estados de agregación de la materia. ¿En cuál sus partículas están siempre juntas dándole una forma definida?
A) Sólido B) Líquido C) Gas D) Plasma
2. El estado de agregación sólido (hielo) del agua, se diferencia del líquido (agua) y del gaseoso (vapor) debido a que:
A) Las partículas que lo forman están ligeramente separadas y toman la forma del recipiente que las contiene.
B) Las partículas están totalmente separadas y no tienen fuerza.
C) Las partículas están estrechamente unidas y le proporcionan forma propia.
D) Sus partículas se expanden fácilmente y chocan con fuerza sobre las paredes del recipiente que las contiene.
3. Imagina que calentamos un trozo de hielo que está a una temperatura inicial de -60°C. Cuando la temperatura del hielo llegue a 0°C, se empezará a derretir. De seguir calentando el agua, sus moléculas se:
A) Desaparecen B) Adhieren C) Comprimen D) Separan
4. ¿Cuál de las siguientes propiedades de los gases es correcta y se manifiesta en un globo aerostático?
A) La velocidad de sus moléculas aumenta cuando la temperatura sube y disminuye cuando baja.
B) Las moléculas de un gas se mueven constantemente, su velocidad es baja y su trayectoria es recta.
C) Las moléculas chocan continuamente unas con otras y contra las paredes del recipiente, perdiendo energía cinética.
D) Las moléculas están juntas unas con otras, el volumen individual es mucho mayor en comparación con el volumen total del gas.
5. Propiedad que caracteriza a las sustancias líquidas.
A) Adquieren la forma del recipiente que los contiene
B) Conserva siempre su forma.
C) Su volumen cambia según el recipiente que los contiene.
D) Se puede romper.
6. Raúl y su equipo se usaron el modelo de partículas y se apoyaron en las aportaciones de Newton y Boltman en el desarrollo del modelo cinético de partículas de acuerdo a dos aspectos básicos:
A) El movimiento y las colisiones entre las partículas de los gases.
B) El movimiento y el vacío que se forma en el cambio de estado de los gases.
C) La forma y el volumen del recipiente que contiene a los gases.
D) La densidad de las partículas de los gases y el volumen del recipiente que los contiene.
7. Mario colocó un globo en un vaso con agua caliente y éste se infló porque:
A) Las partículas de vapor de agua caliente se mueven lentamente.
B) Sólo existen partículas de aire moviéndose dentro en el globo.
C) Se infla por el movimiento de partículas de aire caliente y vapor de agua.
D) Las partículas del vapor de agua se separan y forman un vacío.
8. Haz un dibujo del acomodo de las partículas en cada estado de la materia.
9. Describe el movimiento de las partículas en los 3 estados (Sólido, Líquido y Gaseoso)
10. A diferencia de los sólidos, un gas...
A) Mantendrá su volumen en diferentes contenedores
B) Mantendrá su forma en diferentes contenedores
C) Se expandirá hasta llenar el espacio disponible.
D) Disminuirá su volumen cuando su temperatura se eleve.










No hay comentarios:
Publicar un comentario